Abstract
Introduction: In NPM1-mutated (NPM1mut) acute myeloid leukemia (AML) the detection of measurable residual disease (MRD) using quantitative PCR methods for NPM1mut allows for risk stratification and therapy adjustments during disease course. Even though it is well understood that a persistent NPM1mut burden at allogeneic stem cell transplantation (HSCT) is associated with relapse and shorter survival, the quantitative impact of NPM1mut at HSCT has not been investigated in detail.
Methods: We analyzed 81 NPM1mut AML patients (pts) who received their first allogeneic peripheral blood HSCT at a median age of 61 (range 33-76) years in morphologic complete remission (CR) or CR with incomplete peripheral recovery (CRi). Conditioning regimens were non-myeloablative (70%), reduced-intensity (11%), or myeloablative (19%) according to EBMT guidelines. At diagnosis, cytogenetics and co-mutational status of FLT3, IDH1, IDH2, DNMT3A, and CEBPA were assessed. NPM1 MRD up to 28 days prior to HSCT in CR/CRi was measured by digital droplet PCR using a competitive probe approach, and results were normalized to ABL1 (limit of detection 0.001%NPM1mut/ABL1). For patients with a positive NPM1 MRD test result, receiver operator characteristics (ROC) curves were used to analyze distinct NPM1/ABL1 MRD threshold. Presence of chronic graft-versus-host disease (GvHD) was assessed according to Glucksberg criteria. Median follow up after HSCT was 4.4 years.
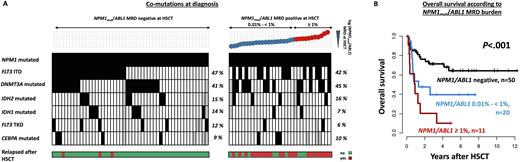
Results: ROC curves identified 0.01% NPM1mut/ABL1 as the most informative cut-off to define MRDpos and MRDnegpts in both, bone marrow and blood, and irrespective of conditioning intensity. Using this cut-off 31 (38%) pts were NPM1mut MRDpos at HSCT and had a significantly higher cumulative incidence of relapse (CIR, P<.001, after 2 years 4% vs 66%) and shorter overall survival (OS, P<.001, after 2 years 77% vs 38%). Patients transplanted NPM1mut MRDpos had a 20.8-times higher risk of relapse than those transplanted MRDneg. While there were no differences regarding the gene mutation profile at diagnosis in pts with or without NPM1mut MRD at HSCT (Figure 1A), pts with NPM1mut MRD at HSCT had a significantly higher diagnostic WBC (49.6 vs 17.9; P=.03), LDH (12.9 vs 6.7; P=.02), and more CD34+ cells in bone marrow at diagnosis (1.4% vs 0.5%; P=.02).
We observed a negative correlation of the NPM1mut /ABL1 MRD levels before HSCT and the time to relapse; the higher the NPM1mut MRD burden at HSCT the earlier the relapse occurred (R= -0.5, P=.03). This correlation was strongest in pts with a FLT3-ITD present at diagnosis (R= -0.82, P=.002). When utilizing two cut-offs for low (≥ 0.01% but < 1%) and high (≥ 1%) NPM1mut /ABL1 MRD levels there was a significantly shorter time to relapse in relapsing AML pts with high NPM1mut MRD levels (median time to relapse: 73 days) before HSCT compared to pts with low MRD levels (median time to relapse: 149 days). In addition, compared to patients with low NPM1mut MRD levels, pts with high NPM1mut MRD levels showed a trend for shorter OS (P=.10; OS after 2 years: 77% for NPM1mut MRDnegpatients vs 48% for low NPM1mut MRDpos patients vs 21% for high NPM1mut MRDpos patients; Figure 1B), indicating that pts transplanted with higher NPM1mut MRD may be harder to salvage. Presence of a FLT3-ITD and age at HSCT did not alter the negative impact on outcomes of low or high NPM1mut MRD at HSCT.
We also evaluated the prognostic impact of the presence of chronic GvHD as a surrogate marker for graft-versus-leukemia effects with respect to the NPM1mut MRD status at HSCT. In a landmark analysis for pts surviving longer than 100 days, NPM1 MRDneg (< 0.01% NPM1mut/ABL1) pts had the lowest CIR (4% after 2 years), followed by NPM1mut MRDpos pts developing a chronic GvHD (25% after 2 years) and by NPM1 MRDpos pts who did not develop a chronic GvHD and had a very high CIR (92% after 2 years, P<.001).
Conclusion: AML patients with > 0.01% NPM1mut/ABL1 MRD before allogeneic HSCT had a significant higher risk of relapse and shorter OS. High WBC, LDH, and CD34+ cells in bone marrow at diagnosis associated with persisting NPM1mut MRD at HSCT. The higher the NPM1mut MRD burden at HSCT, the earlier the relapse, and the lower the chances of successful salvage therapy after relapse. Development of a chronic GvHD after HSCT was associated with a reduced risk of relapse in NPM1mut MRDpos pts, indicating that bolstering immunologic effects post-HSCT for NPM1mut MRDpos AML pts may be a promising option to improve outcomes.
Disclosures
Schwind:Novartis: Honoraria. Herling:Abbvie: Honoraria, Research Funding; EDO-Mundipharma: Honoraria, Research Funding; Janpix: Honoraria, Research Funding; Jazz: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Roche: Honoraria, Research Funding; Takeda: Honoraria, Research Funding. Merz:BMS Celgene: Honoraria; Janssen: Honoraria. Metzeler:Daiichi Sankyo: Honoraria; Pfizer: Consultancy; Jazz Pharmaceuticals: Consultancy; Novartis: Consultancy; Celgene/BMS: Consultancy, Honoraria, Research Funding; Curis: Research Funding; Astellas: Honoraria; AbbVie: Honoraria. Franke:Novartis: Honoraria; Jazz Pharmaceuticals: Honoraria; Pfizer: Honoraria; Gilead: Other: Travel support; Incyte: Honoraria; BMS: Honoraria; Takeda: Other: Travel support. Vucinic:Novartis, Gilead Kite, Takeda, MSD, BMS Celgene, Abbvie, Amgen: Honoraria; MSD, BMS Celgene, Novartis, Gilead Kite, Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees; Sobi, BMS Celgene: Other: travel, accommodations, expenses. Jentzsch:Novartis: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal